Category: Featured News
Five-Year Study Confirms Lasting Benefits of Immunotherapy for Mesothelioma
Key findings
- Five years after treatment, 14 percent of patients who received immunotherapy were alive, compared with 6 percent of those treated with chemotherapy.
- Among patients with epithelioid mesothelioma, five-year survival reached 14 percent with immunotherapy versus 8 percent with chemotherapy.
- For patients with non-epithelioid mesothelioma, five-year survival was 12 percent with immunotherapy and only 1 percent with chemotherapy.
- Median overall survival was 18.1 months with nivolumab plus ipilimumab, compared with 14.1 months for chemotherapy.
- Tumor response occurred in about 39 percent of patients receiving immunotherapy and 44 percent receiving chemotherapy.
- Seventeen percent of immunotherapy responders still had an ongoing response five years later.
A newly released five-year analysis from the landmark CheckMate 743 clinical trial provides encouraging evidence that immunotherapy can produce long-term survival for some people with unresectable pleural mesothelioma. The study results, highlighted by oncology news outlet OncoDaily and published in the Journal of Clinical Oncology, show that a combination of two immune checkpoint inhibitors continues to outperform traditional chemotherapy over time.
Pleural mesothelioma is a rare cancer that develops in the lining of the lungs and is strongly associated with asbestos exposure. For many years, treatment options were limited and long-term survival was uncommon. The updated results from CheckMate 743 suggest that immunotherapy is helping to change that outlook for some patients.
Comparing immunotherapy with chemotherapy
The CheckMate 743 trial evaluated the combination of nivolumab and ipilimumab as a first-line treatment and compared it with the standard chemotherapy regimen of pemetrexed (Alimta) together with cisplatin or carboplatin. Researchers tracked patient outcomes for a median follow-up period of nearly 67 months, providing the longest observation period yet for a first-line immunotherapy approach in pleural mesothelioma.
Investigators reported that the immunotherapy combination continued to demonstrate a survival advantage. As the researchers wrote in the journal article, “nivolumab plus ipilimumab demonstrated continued overall survival benefit versus chemotherapy in all randomly assigned patients.”
At the five-year mark, 14 percent of patients who received the immunotherapy regimen were still alive. In contrast, only 6 percent of patients treated with chemotherapy reached that milestone. The hazard ratio for overall survival was 0.74, meaning the risk of death was reduced by roughly 26 percent in the immunotherapy group.
Survival differences by tumor type
Mesothelioma is often categorized by histology, or tumor type. The most common form, epithelioid mesothelioma, typically responds somewhat better to treatment than the more aggressive non-epithelioid forms.
In the updated analysis, patients with epithelioid tumors experienced a five-year survival rate of 14 percent when treated with nivolumab plus ipilimumab, compared with 8 percent among those who received chemotherapy.
The contrast was even more pronounced in patients with non-epithelioid disease. In this group, five-year survival reached 12 percent with immunotherapy but only 1 percent with chemotherapy. The hazard ratio for overall survival among these patients was 0.48, suggesting a substantially lower risk of death when the immunotherapy combination was used.
Median survival and long-term outcomes
Median overall survival, which reflects the point at which half of patients are still alive, was 18.1 months for those treated with nivolumab plus ipilimumab. Patients who received chemotherapy had a median survival of 14.1 months.
Although these figures may appear modest, they represent progress in a cancer that has historically been difficult to treat. Median survival does not represent an average life expectancy. Instead, it marks the midpoint of the survival curve. Many patients may live longer than that value, and some significantly longer.
The five-year survival rate seen with immunotherapy is particularly notable given the historically low rates of long-term survival in mesothelioma.
Tumor responses and durability
Initial response rates were relatively close between the two treatment approaches. About 39 percent of patients receiving nivolumab plus ipilimumab experienced tumor shrinkage, compared with 44 percent of those treated with chemotherapy.
However, the durability of those responses differed dramatically.
Among patients whose tumors responded to immunotherapy, 17 percent continued to show a response five years later. In the chemotherapy group, none of the responders maintained an ongoing response at the five-year mark.
This pattern suggests an important distinction between the treatments. Chemotherapy may produce faster tumor shrinkage in some patients, but immunotherapy appears capable of generating longer-lasting control in a subset of cases.
Investigating potential biomarkers
Researchers also explored whether certain blood-based indicators might help predict which patients benefit most from immunotherapy. One area of focus involved monocytic myeloid-derived suppressor cells, often abbreviated as M-MDSCs.
These immune cells are believed to interfere with the body’s anti-cancer immune response and may limit the effectiveness of immunotherapy. The study found that patients who had higher levels of M-MDSCs at the start of treatment tended to experience poorer overall survival when treated with nivolumab plus ipilimumab.
Specifically, the hazard ratio for overall survival was 1.25 among patients with higher baseline levels compared with those who had lower levels. Researchers emphasized that this analysis is exploratory and will require additional study before such markers could guide treatment decisions.
Accounting for treatment crossover
Another factor examined in the analysis involved patients who switched treatments during the study. About 24 percent of participants who initially received chemotherapy later went on to receive immunotherapy.
To better understand the true effect of starting immunotherapy first, investigators performed an adjusted statistical analysis that accounted for this crossover. After making that adjustment, the hazard ratio for overall survival improved to 0.64 in favor of nivolumab plus ipilimumab.
The adjusted analysis also reduced the estimated median survival for the chemotherapy group to 12.1 months, further emphasizing the benefit associated with beginning treatment with immunotherapy.
Safety findings remain consistent
The extended follow-up period did not reveal any new safety concerns. According to the study authors, “no new safety signals were observed” during the longer observation period.
Immune checkpoint inhibitors can still cause immune-related side effects, which may affect organs such as the lungs, liver, or thyroid. However, the safety profile reported in the five-year update was consistent with what had been observed earlier in the trial.
A meaningful step forward
In their concluding remarks, the investigators noted that the updated data show sustained, long-term clinical benefits from nivolumab plus ipilimumab when used as a first-line treatment for pleural mesothelioma. The survival advantage was observed regardless of tumor histology.
As a result, the findings further reinforce the role of the immunotherapy combination as a standard treatment option for patients with unresectable pleural mesothelioma.
For individuals and families facing this diagnosis, the results do not represent a cure. Still, the ability of a therapy to help a portion of patients live five years or longer is a meaningful advance in a disease where durable survival has historically been rare.
Ongoing research will continue to explore how immunotherapy can be improved, identify which patients benefit most, and determine how new treatment strategies might build on the progress already achieved.
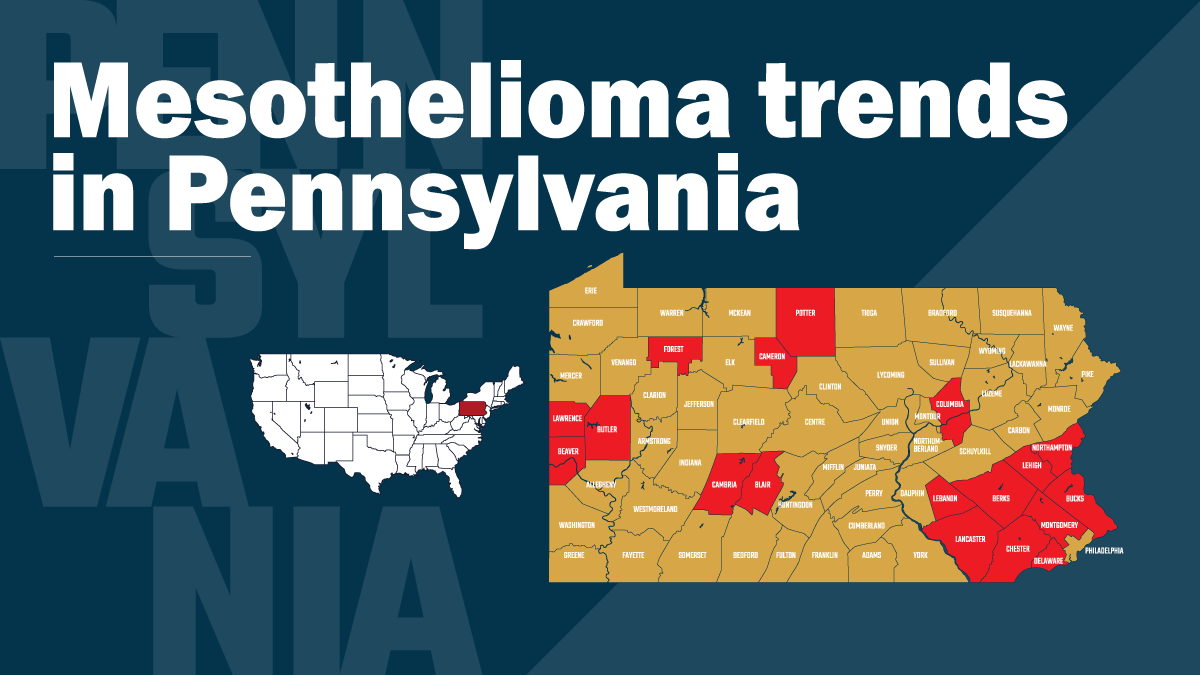
How mesothelioma rates changed in Pennsylvania over three decades
A recent study looked at nearly 30 years of mesothelioma data in Pennsylvania, using records from the Pennsylvania Cancer Registry covering 1990 through 2019. The research gives a clearer picture of how this rare cancer has changed over time, how it varies by type, and where within the state it has been most common.
What the data shows
Mesothelioma is a rare cancer that forms in the lining of organs such as the lungs or abdomen. It is most often linked to past asbestos exposure and usually shows up many years after contact with asbestos fibers.
Researchers reviewed about 6,000 confirmed cases from the registry. They focused on the two most frequent forms of the disease:
Pleural mesothelioma: affects the lining of the lungs
Peritoneal mesothelioma: affects the lining of the abdomen
In the dataset, around 85 percent of cases were pleural type and fewer than 10 percent were peritoneal, reflecting national patterns.
Pleural mesothelioma changing trends in Pennsylvania
Pleural mesothelioma cases climbed steadily through the 1990s and 2000s, then began to fall around 2005. The authors link this decline to stronger workplace asbestos regulations from earlier decades, with benefits appearing only after the long delay that is typical for this disease.
Peritoneal mesothelioma changing trends in Pennsylvania
Peritoneal mesothelioma stayed rare throughout the 30 years studied. Overall numbers did not change much, but there was a noticeable increase among younger adults and women. This may point to risk factors beyond traditional workplace exposure, though more research is needed.
Where is mesothelioma most common
Mapping the data showed geographic patterns in both types of mesothelioma. Higher rates of pleural cases appeared in several southeastern and western counties, areas with a history of heavy industry and asbestos use. Peritoneal mesothelioma showed a broader, more scattered distribution across the state, with some overlap in the same regions.
Why this matters
Even decades after the peak use of asbestos in industry, health effects are still showing up. The trends for pleural mesothelioma likely reflect past occupational exposures, while trends for peritoneal mesothelioma raise questions about other exposure routes and detection patterns. Researchers emphasize the need for ongoing monitoring and better understanding of all forms of asbestos exposure, especially in communities with higher case rates.

The Unseen Impact of Mesothelioma on Family Caregivers
A new study looking at how caregivers of mesothelioma patients experience the disease journey has released its first set of findings. The research, published in the Innovation in Aging journal in December 2025, focuses specifically on the roles and challenges of family members who provide care from diagnosis through bereavement.
Mesothelioma is a rare cancer caused by exposure to asbestos. It often progresses quickly and brings complex emotional, physical, and practical challenges for both patients and their families. Historically, studies tend to focus mainly on patients. This new research is among the first to put caregivers at the center, acknowledging the intense demands placed on them as they support a loved one while balancing their own lives.
The initial phase of the study involved interviews with caregivers, exploring their real-world experiences. Early results show that caregivers face overlapping forms of stress.
Emotional strain and uncertainty were common themes. Many caregivers spoke about the difficulty of coping with not knowing what to expect after a mesothelioma diagnosis, especially given the disease’s aggressive nature.
The day-to-day responsibilities often go far beyond medical support. Caregivers described managing treatment plans, coordinating medical appointments, handling medications, and balancing household and career duties. These practical demands can be exhausting and relentless.
The psychological toll was another major concern. Caregivers reported significant stress and anxiety not only from watching a loved one’s health decline but also from navigating complicated legal, financial, and healthcare systems tied to mesothelioma.
Importantly, the research highlighted that caregivers’ experiences do not end when the patient dies. Many participants described feeling unprepared for bereavement and unsupported once they were no longer immersed in the healthcare system they had relied on during caregiving.
Researchers say these early findings point to the need for greater recognition of caregiver experiences and more long-term support tailored to their needs. As research continues, it may help shape better care models that account for both patients and the family members who care for them.
The study, led by researchers at Michigan State University, is currently open to caregivers of mesothelioma patients. To inquire about eligibility, visit https://msu.co1.qualtrics.com/jfe/form/SV_b7LKdErA70374zA.

New Free Clinical Trials Search Tool for Mesothelioma Launches
A new, free online search tool dedicated exclusively to mesothelioma clinical trials has launched on mesotheliomahelp.org, offering patients, physicians, and researchers a focused resource to find and share trials specific to mesothelioma and its subtypes. The platform, which is the first of its kind devoted solely to mesothelioma, combines detailed disease-specific filters with geographic search capability and an easy process for clinicians and investigators to submit trials for inclusion.
The tool’s design focuses on two core needs: helping patients and families find relevant trials quickly and giving doctors and researchers a simple way to make their trials visible to a larger, targeted audience. Users can search by type of cancer (for example, pleural mesothelioma, peritoneal mesothelioma, tunica vaginalis mesothelioma, or pericardial mesothelioma) and then narrow results by geographic location, from state-level searches down to city or zip-code proximity options. The combination of disease specificity and location-based searching is meant to cut through the noise of broader clinical-trial registries and accelerate connections between patients and trials that are realistically accessible.
Search Clinical TrialsOnce a patient or caregiver builds a shortlist of promising trials, the site emphasizes the practical next step: patients are encouraged to take the list to their treating physicians to discuss feasibility and eligibility. The goal is to put the patient in the driver’s seat but with a solid reminder that treatment should be a collaboration between patient/family and physician. Having a curated list of clinical trials in hand at their appointment can help streamline the conversation and rapidly assess whether a trial’s inclusion criteria apply to that patient based on data from previous phases of that trial, the patient’s disease stage, previous treatment and response, and wellbeing.
The platform also invites doctors and researchers to submit their trials for inclusion.
While clinical trials are experimental and not yet proven treatments, in a rare cancer like mesothelioma with few available treatments with limited effectiveness, they are a way to try something different, especially when other things haven’t worked or stopped working. But that doesn’t mean that no data exists for these novel treatments. In fact, the opposite is often true. The new treatments tend to already have data in other cancers based and if the study is a Phase 3 clinical trial, then it also has data signals for mesothelioma specifically.
Clinical trials proceed in phases — from early, small safety studies (Phase I) to larger tests of effectiveness (Phase II and III) and post-approval follow-ups (Phase IV). For patients with mesothelioma, particularly those with advanced or treatment-resistant disease, clinical trials can provide access to the latest therapies before they are widely available, including targeted therapies, immunotherapies, and novel combination strategies. Participation can offer potential clinical benefits such as tumor shrinkage or prolonged disease control; at the same time, trials also carry risks and may not be effective for every participant. Importantly, clinical trials generate the evidence that advances cancer treatment for all patients in the future.
The site’s mesothelioma-only focus is the feature that sets it apart. Whereas registries that catalog trials across all cancers can be comprehensive, they are not always user-friendly for users without a medical background. By concentrating on one disease group, the search tool can present deeper, more relevant filtering options and educational material tailored to mesothelioma patients’ unique needs.
For investigators and research sites, the platform also offers visibility benefits. Submitting a trial to the search tool increases the chance that eligible patients and referring physicians who are specifically searching for mesothelioma options will find the trial. That improved visibility can accelerate enrollment, reduce screening times, and help trials reach target accrual sooner.
As clinical research continues to expand and treatments for mesothelioma evolve rapidly, tools that connect patients and clinicians to appropriate studies are increasingly valuable. This mesothelioma-dedicated, free, searchable database with disease-type and location filters and a direct submission process for investigators aims to be a practical bridge between the clinical-research community and the people who may benefit from participation. Patients and clinicians interested in exploring the new search tool can visit mesotheliomahelp.org to begin searching, learn how to submit trials, and find resources to guide conversations with treating physicians about clinical-trial options.

What Experts Revealed at iMig 2025 About the Next Wave of Mesothelioma Therapies
The 2025 gathering of the International Mesothelioma Interest Group (iMig) offered a powerful snapshot of where treatment for mesothelioma is heading, with a mix of clinical debate, scientific breakthroughs, and evolving therapeutic strategies. Several recurring themes emerged, notably around the role of surgery, advances in targeted therapies, and the expanding use of systemic treatments.
Surgery Under Scrutiny, but Still an Option in the United States
One of the central conversations revolved around the findings of the MARS2 trial, a UK-based multicenter study that randomized pleural mesothelioma patients (after two cycles of chemo) to either continue chemotherapy alone or undergo surgery followed by further chemo. The trial concluded that surgery provided no clear survival benefit.
These results shook long-held assumptions in the field and stimulated a strong re-evaluation of when surgery should be used. Experienced surgeons at the conference argued that the trial may not reflect optimal, real-world surgical practice citing concerns about patient selection and higher mortality in the surgical group.
Still, some high-volume mesothelioma centers (especially those handling 25+ cases per year) maintain that surgery can remain part of a broader multimodal treatment plan. In other words: though surgery may not cure, it could help extend life when combined with modern systemic therapies performed at experienced centers.
Because current systemic treatments yield a median survival around 18–24 months, combining refined surgical strategies with systemic therapy remains a goal. Trials incorporating immunotherapy either before or after surgery were spotlighted as especially promising.
A New Target: The Hippo Pathway and TEAD Inhibitors
Beyond surgical debates, researchers pointed to new molecular avenues such as the dysregulated Hippo pathway, which is disrupted in roughly 60% of mesothelioma cases. This makes it an attractive target for drug development.
Agents like Vivace VT3989 (a TEAD palmitoylation inhibitor) and IAG933 (which interferes with TEAD-YAP/TAZ interactions) are currently under development or in regulatory review for advanced solid tumors, including mesothelioma.
Preclinical data presented at iMig suggest that combining TEAD inhibition with immunotherapy may enhance immune cell infiltration into tumors, potentially improving effectiveness. However, the presence of Hippo pathway abnormalities alone isn’t yet a reliable predictor of who benefits.
This line of research could open new doors for patients, especially those whose tumors aren’t well served by existing chemo or immunotherapy regimens.
Systemic Therapies
Attendees reviewed the current arsenal of systemic treatments and looked ahead to future combinations. For patients who relapse, single-agent immunotherapy remains an option: for example, nivolumab has demonstrated improved progression-free survival compared with placebo, though overall survival gains are modest.
Other agents, such as pembrolizumab, show response rates in relapsed disease similar to older chemo drugs like gemcitabine or vinorelbine.
More importantly, several clinical trials are now exploring combinations intended to improve outcomes. For instance:
- The BEAT‑meso trial is testing standard chemo plus a blend of an immunotherapy agent and a VEGF inhibitor.
- The IND 227 trial compared chemo alone to chemo plus pembrolizumab, revealing particular benefit for non-epithelioid subtypes such as sarcomatoid or biphasic mesothelioma.
- Other trials include MIST 4 trial (atezolizumab + bevacizumab after prior chemo), RAMES trial (gemcitabine with ramucirumab vs. placebo), and investigations of antibody-drug conjugates like anetumab ravtansine, either alone or combined with immunotherapy.
New Hope for Aggressive Subtypes: Sarcomatoid and Biphasic Mesothelioma
Patients with sarcomatoid or mostly sarcomatoid tumors have long faced grim prognoses, given that those subtypes respond poorly to standard treatments. Results from studies in recent years, though, have shown growing evidence that immunotherapy can meet a critical need here.
One particularly important development: in nonepithelioid pleural mesothelioma, the combination of chemotherapy with pegargiminase significantly improved outcomes in the ATOMIC‑meso trial. According to the data discussed, pegargiminase caused a modest increase in overall survival (about 1.6 months), but more strikingly, quadrupled 3-year survival compared to chemotherapy alone. The drug works by starving tumor cells of arginine, a nutrient certain mesothelioma cells can’t produce on their own, which weakens their growth ability.
While immune-based regimens remain powerful, they can also lead to unpredictable side effects; in such cases, pegargiminase represents a promising alternative or addition for individuals who may not tolerate immunotherapy or who are not candidates for immunotherapy.
What’s Next
Discussions at iMig underscored a balance between realism and optimism. On one hand, the field recognizes the many hurdles left to overcome as mesothelioma remains difficult to treat, and no single approach is a silver bullet. On the other, there is clear momentum toward better patient outcomes, built on improved surgical techniques, deeper biological understanding, novel drug targets, and smarter combination therapies.
The conference reaffirmed that the future of mesothelioma care likely lies in personalization: matching treatment strategies (surgery, chemo, immunotherapy, targeted agents) to tumor biology, disease subtype, and patient condition. With ongoing clinical trials and evolving research, there is growing hope that the standard of care will continue to shift giving patients more options and, ultimately, much more time.
Free Mesothelioma Patient & Treatment Guide
We’d like to offer you our in-depth guide, “A Patient’s Guide to Mesothelioma,” absolutely free of charge.
It contains a wealth of information and resources to help you better understand the condition, choose (and afford) appropriate treatment, and exercise your legal right to compensation.
Download Now